 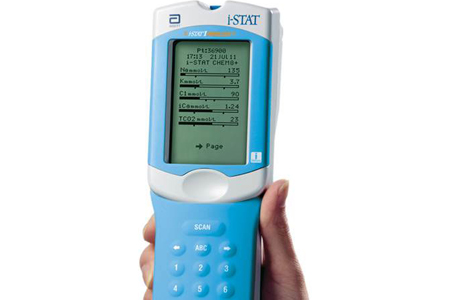
The ACT was measured simultaneously with the other method. I hope the release of this preliminary data will prompt other clinical laboratories to review their results. Methods: We randomized 97 patients undergoing coronary artery bypass grafting or aortic valve replacement to either kaolin- or Celite-guided anticoagulation. I have decided to discontinue bedside INR testing with ISTAT until this discrepancy has been resolved. Prothrombin time (PT) and partial thromboplastin time (PTT) are tests used to evaluate coagulation, or the clotting of blood. Whole blood analysis forms the mainstay of perioperative coagulation point-of-care testing (POCT) allowing insight into the complex interactions within the clotting system, reducing turnaround test times, and helping minimize inappropriate transfusion. Clay particles (kaolin) activate the intrinsic clotting pathway leading to activated thrombin, which acts on a synthetic thrombin substrate forming an electrochemically active product. Creatinine measurements are used in the diagnosis and treatment of renal diseases, in monitoring renal dialysis, and as a calculation basis for measuring other urine analytes. Preanalytical variables in coagulation testing: setting the stage for accurate results. The i-STAT Creatinine test is used for the quantitative determination of creatinine (crea) in whole blood on the i-STAT handheld. In UFH treated blood, iSTAT and Hemochron showed a significant change of ACT at 0.075 IU/ml and 0.1 IU/ml UFH. This small single study suggests that clinicians cannot make accurate clinical decisions based on I-STAT INR results. whole blood coagulation test similar to the activated partial thromboplastin time (PTT) test using plasma. Simultaneous ACT measurements were performed using iSTAT and Hemochron. These letters mean that the CG4+ (BLUE) and G3+ (BLUE) are now considered high complexity tests due to lack of FDA categorization. ISTAT INR was as much as 0.5 lower than STAR when the STAT INR is 2.8.This study demonstrated significant INR differences between I-STAT and STAR. These results were much more discrepant than the results published in the I-STAT Prothrombin Time/PT packgage insert that compared I-STAT to STA Compact. Even more disconcerting was that the bias was inconsistent throughout the therapeutic range. The majority of the studies involve Hemochron, Medtronic or i-STAT devices. Recently, I compared the INR results of patients treated with warfarin who were simultaneously tested with a fingerstick specimen on an I-STAT at the bedside and a venipuncture specimen on a Stago STAR coagulation analyzer located in the main clinical laboratory. Presently the activated clotting time (ACT) remains the primary test for. I-STAT INR cartridges are used to determine if a patient presenting to the ED with an ischemic stroke can receive tissue plasminogen activator or if a patient anticoagulated with warfarin can undergo an emergent invasive procedure. I-STAT (Abbott Point of Care) is a popular device for point of care testing that is used in hospitals to provide quicker turnaround time and expedite clinical decision making.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
